Ramsay’s scientific works related to the Brownian movement dealt a crushing blow to the opponents of atomism and made all scientists finally convinced that atoms and molecules exist. This is hard to believe, but some scientists denied the atomic-molecular theory and did not believe in the existence of molecules and atoms even at the beginning of the 20 century. Just as the waves swinging a distant boat are not visible from the shore, although the movement of the boat itself is visible.īrownian motion became one of the important components of atomic-molecular theory and at the same time an important proof of the fact that all matter consists of the smallest particles: atoms and molecules. 
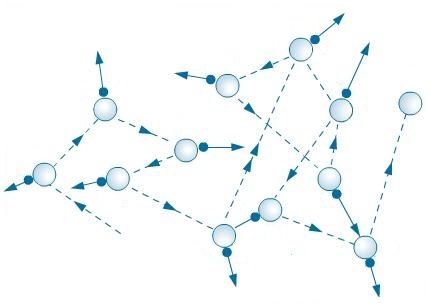
He managed to prove that the cause of the Brownian motion of small particles is the impact of even smaller particles, which are no longer visible in a normal microscope. The famous Scottish physicist and chemist William Ramsay was among them. Later, Wiener’s ideas were developed by other scientists. This was the first explanation of this strange phenomenon, associated with the properties of atoms and molecules, the first attempt to reveal the secret of the structure of matter using Brownian motion. In 1863, the German mathematician Ludwig Christian Wiener suggested that Brownian motion happens due to the vibrational movements of certain invisible atoms. Its explanation was given only at the end of the 19 century, and it was not immediately accepted by the scientific community. Moreover, the Brownian movement was completely random and did not succumb to any logic. However, scientists could not explain the reasons for the Brownian movement for many years. Robert Brown was happy to show it to many of his colleagues. Atomic-molecular Theoryīrownian Motion soon became very famous in scientific circles. If you’ll look through this microscope on a blackened box with smoke and illuminated by a wide beam of light, you’ll see small pieces of soot and ash that will continuously jump back and forth. This is interesting: you can observe the Brownian motion with your own eyes, for this you’ll need a strong microscope (after all, there were no powerful modern microscopes during the life of Robert Brown). These “active molecules” were observed everywhere, being in constant and chaotic motion. Then glass, various and various minerals came under the microscope of the researcher. The scientist was even more surprised when he began to study inanimate matter: small particles of coal, soot, and even dust particles of London air. But particles of dead plants (and even plants dried up a hundred years ago in herbaria) had a similar movement. Robert Brown, discoverer of the Brownian movement.Īt first, Robert Brown thought that he was observing the movement, even the “dance” of some living microorganisms, because the pollen itself is, in fact, the male reproductive cells of plants. In particular, it was noted that particles tend to accelerate with increasing temperature, as well as with decreasing particle size. This observation was confirmed by other scientists. One day, observing the movement of pollen in vegetable juice in a microscope, the scientist noticed that small particles make random tortuous movements. He was particularly interested in how pollen takes part in plant propagation. In 1827, Robert Brown was engaged in active research on pollen from various plants. It is in his honor it was called “Brownian”. The discoverer of the Brownian movement was the English botanist Robert Brown (1773-1858). What is the story of the discovery of Brownian motion? Why is Brownian motion so important in physic and chemistry? What are some examples of Brownian motion in real life? Read about all this in our article. The cause of Brownian motion is the collision of small particles with other particles. Brownian motion (or Brownian movement) is the chaotic and random motion of small particles (usually molecules) in different liquids or gases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
